Artificial foldamers: Design, synthesis and characterization
Our group has developed a unique know-how in the design, synthesis and characterization of artificial folded molecular architectures (foldamers) based on aromatic oligoamides. These objects differ notably from peptides or nucleotides, from which they are inspired, and from their analogues, and give access to structural motifs and thus to functions beyond those of biopolymers.
Theme 1
Foldamer design
The helix, turn, and β-strand motifs of biopolymer folded structures have been found to prevail also in non-natural backbones. In contrast, foldamers with aryl rings in their main chains possess distinct conformations that may give access to folded objects beyond the reach of peptidic and nucleotidic backbones. Therefore the functions featured by artificial backbones would eventually go beyond those of natural backbones. The aromatic oligoamide foldamers that our group developed have proven to be remarkable objects in that their folded conformations are extremely stable, tunable and easy to predict. Their use can span from the development of proteomimetics to fundamental investigations on folding phenomena. They can also be used in molecular recognition using a unique design of helical molecular containers that completely surround small organic guests (see below). Water soluble foldamers also show highly promising biological properties, especially for their interactions with proteins (see below). At last, the thermodynamic stability of helical conformations was determined, and peculiar chemical and physical behaviors were demonstrated paving the way towards new developments. For example, it was shown that aromatic helices mediate electron transfer between donors and acceptors at their extremities.
Theme 2
Molecular Recognition within foldamers
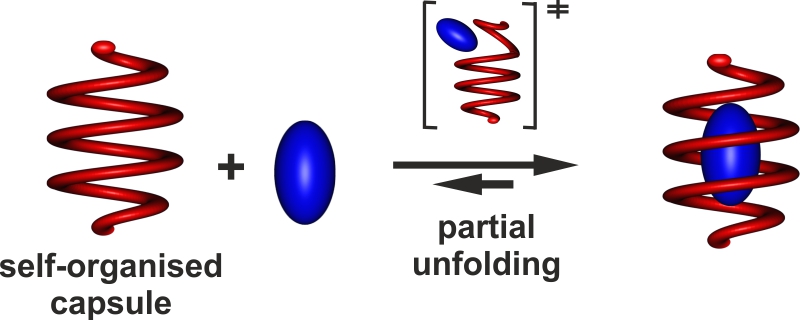
The ab initio rational structure-based design of a synthetic molecular receptor for a given complex biomolecular guest remains an elusive objective, yet remarkable progress has been achieved in recent years. Our group uses of folded artificial aromatic amide oligomers also termed aromatic foldamers, inspired from biopolymer structures, for the design of helical molecular capsules that can recognize guest molecules, completely surround them and isolate them from the solvent, thus giving rise to a sort of guest encapsulation associated with slow binding and release kinetics. The development of new amino acid, diacid and diamine monomers, a main source of creativity in this field, progress in their assembly into ever longer oligoamide sequences, and the predictability of the folded structures due to their inherent rigidity and simple folding principles, allowed for the design and preparation of unimolecular and bimolecular capsule shapes. These capsules consist of molecular helices having a large diameter in the middle and a narrow diameter at both ends thus creating a cavity suitable for binding a guest molecule.
Theme 3
Biological applications of foldamers
Helical aromatic oligoamide foldamers have been developed in our lab with fully cationic side chains conferring them with water solubility and and cell penetration properties.[1] Advances in synthetic folded architecture design together with the finding that some aromatic backbones also fold in water have opened avenues toward selective foldamer–biomolecule interactions and foldamers that interfere with biological functions.
Contact us
You want to work with us ?
PhD positions are available in our group each year, usually starting in October. If you are interested in doctoral studies in the group then please get in touch as early as possible. An application should consist of your CV, a cover letter, and two reference letters.
Post-doctoral positions: Enquiries are particularly encouraged from applicants with experience in the synthesis and characterisation of macromolecules. Experience in solid phase synthesis would also be appreciated.